The Background
Why is mercury a concern in the Arctic?
What is Mercury?
Mercury is a heavy metal that has long been naturally present in the environment. In its pure form, it is called elemental, or metallic mercury. This is typically the shiny metal used in old thermometers and some electrical switches. This form rarely exists naturally. When combined with elements such as chlorine, sulfur, or oxygen, it forms inorganic mercury compounds or salts, usually white powders or crystals. Finally, when combined with carbon, often through microbial action in aquatic ecosystems, it forms organic mercury compounds such as methylmercury. While all forms of mercury have certain levels of toxicity, methylmercury is the most toxic form.
What are the sources of Mercury?
Mercury in the environment is constantly cycled and recycled as a result of natural and anthropogenic sources and processes, taking different chemical forms along the way. Primary natural sources of mercury are volcanoes, rock weathering, and geothermal activity. Primary anthropogenic sources are the combustion of fossil fuels, smelting activities. Several natural and anthropogenic processes contribute to transport and distribution of mercury through the land-atmosphere-ocean system (see fig. 1).
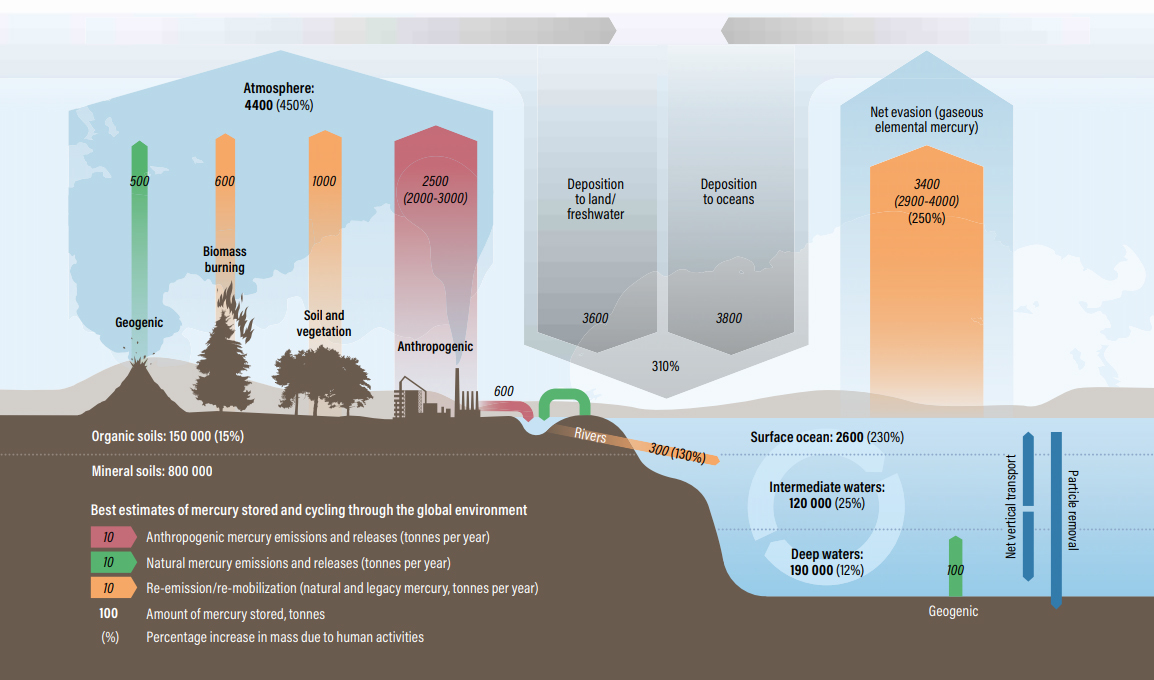
Figure 1: The updated global mercury budget shows the impact of human activities on the mercury cycle and the resulting increase in mercury accumulated in soils and oceans. Source: UN Environment, 2019. Global Mercury Assessment 2018. UN Environment Programme, Chemicals and Health Branch Geneva, Switzerland.
How does mercury make its way to the Arctic?
The Arctic is predominantly influenced by long-range atmospheric and oceanic transport of mercury (see Figure 2). Transport and delivery of mercury to the Arctic is extremely complex, in part due to exchanges and transformations of mercury between air, soil, water and the cryosphere.
It is of particular concern because of its ability to accumulate through time in living organisms, and to build up at each successive level of the food web, exposing wildlife and indigenous populations that consume these organisms as part of a traditional diet.
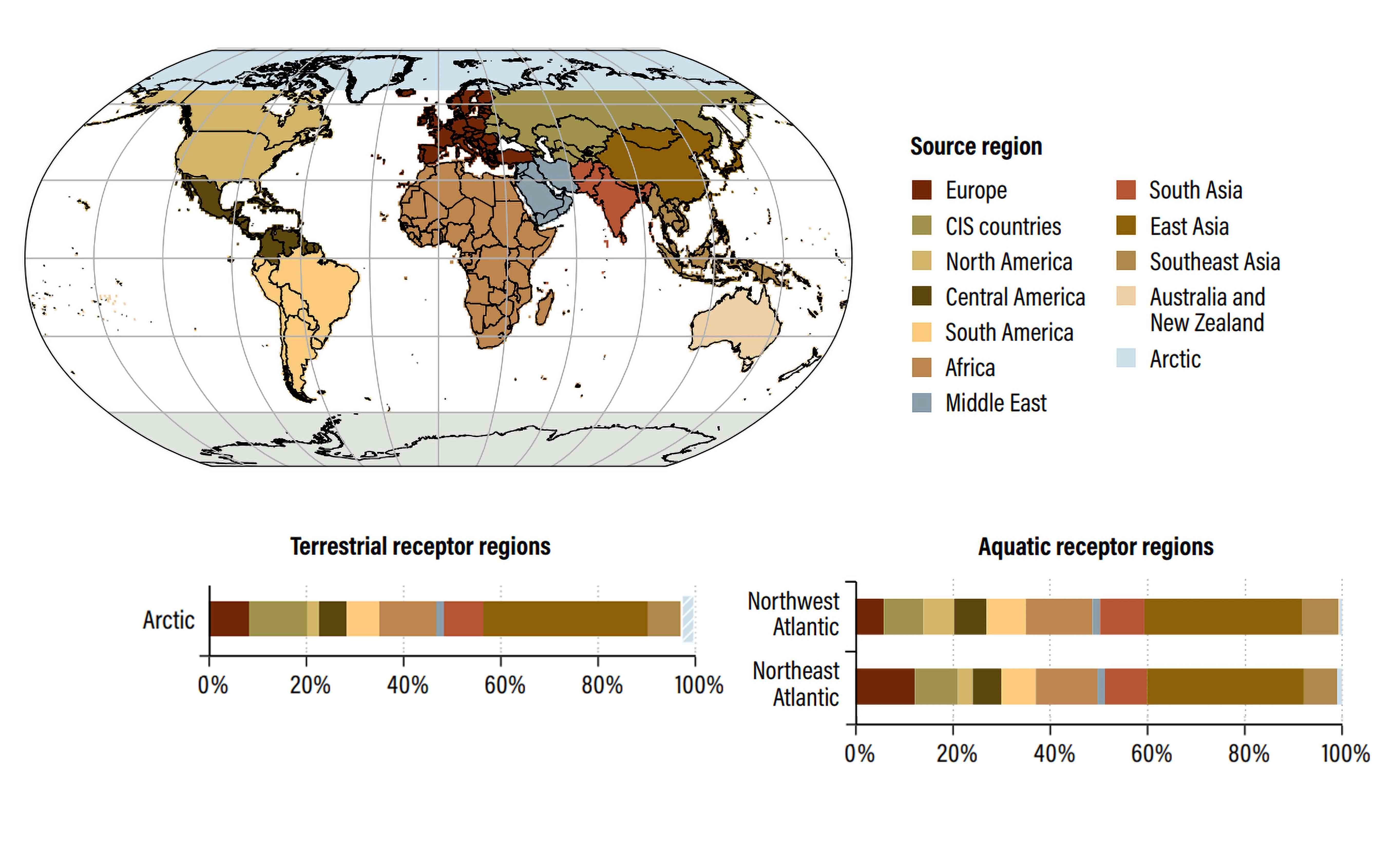
Figure 2: Source apportionment of mercury deposition from anthropogenic emissions to various terrestrial (left) and aquatic regions (right) in 2015. Colors depict source regions, indicated in map above: hatching shows the deposition in a particular region that originates from the region itself. Modified from: UN Environment, 2019. Global Mercury Assessment 2018. UN Environment Programme, Chemicals and Health Branch Geneva, Switzerland.
Figure 3: Model simulation of mercury air transport in 2015 showing characteristic spring to early summer trans-Pacific transport and many other features. Units: modelled elemental mercury vertical column density (VCD) in µg/m2. Credit: Environmental and Climate Change Canada Mercury modeling (Ashu Dastoor and Andrei Ryjkov).
How does Mercury impact Arctic biota?
Microbes and unicellular organisms are thought to be one of the main entry points for methylmercury into food webs. Once assimilated by the lower levels of Arctic food chains, organisms that feed on them will themselves be consumed by higher-trophic level organisms, increasing bioaccumulation at each trophic level. High concentrations of methymercury can affect reproduction of top predators.

How does Mercury impact human populations of the Arctic?
Traditional diets and cultures of human populations in the Arctic are often comprised of wildlife at the top of the food web such as polar bears, beluga whales, seals, reindeer and caribou, which can contain high levels of mercury through processes of bioaccumulation and biomagnification. Mercury can cause neurological damage and hamper the development of children.
Mercury and climate change in the Arctic
The Arctic is currently one of the fastest-warming regions on Earth. Climate change has the ability to alter the transport, distribution, chemical interactions, deposition, and release of mercury in the Arctic regions. This amplifies the need for an understanding of global mercury trends. Studies have revealed that climate change is already having an impact in biota in lower latitudes regions.

